| atomic_structure.ppt | |
| File Size: | 766 kb |
| File Type: | ppt |
What you need to know checklist - The Atom
• Atoms are made of 3 sub-atomic particles called protons, neutrons + Electrons
• Protons have a positive charge
• Electrons have a negative charge
• Neutrons have a Neutral Charge (This means they have no charge)
• They are so small they cannot even be measured in grams, instead they are measured in atomic mass units (a.m.u)
• The proton and neutron have a mass of 1 a.m.u
• The electron has a mass of 1/1,840 a.m.u! This is very tiny!
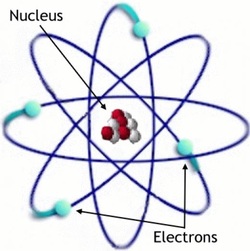
• Protons and neutrons are at the centre of the atom in the nucleus
• Electrons are found in shells called energy levels around the nucleus and whizz around it
• What makes atoms different is the number of protons in the nucleus
• Each atom has its own number of protons in its nucleus
• The number of protons in its nucleus is called its atomic number
• Atoms are neutral because they have the same number of elecrons and protons
• For example if carbon is atom number 6 it has 6 protons and 6 electrons
• The electron configuration tells us how the electrons are arranged in an atom
• The first energy level holds 2 electrons, the second can hold up to 8 electrons, the third can hold up to 8 electrons and so can every other energy level after that
• For example an atom of Nitrogen has 7 electrons, 2 go in the first energy level and there are 5 left over so these go in the next energy level, its electron
configuration is 2, 5
• For example an atom of Potassium has 19 electrons, 2 go in the first energy level and there are 17 left over so 8 of these go in the next energy level, This leaves
9 electrons, 8 of these go in the next energy level which leaves 1 electron this go in the last energy level, its electron configuration is 2,8,8,1
• Some atoms have different numbers of neutrons in their nucleus even though they are the same atom, these are called isotopes
• Isotopes are atoms of the same element with different numbers of neutrons
• Protons have a positive charge
• Electrons have a negative charge
• Neutrons have a Neutral Charge (This means they have no charge)
• They are so small they cannot even be measured in grams, instead they are measured in atomic mass units (a.m.u)
• The proton and neutron have a mass of 1 a.m.u
• The electron has a mass of 1/1,840 a.m.u! This is very tiny!
• Protons and neutrons are at the centre of the atom in the nucleus
• Electrons are found in shells called energy levels around the nucleus and whizz around it
• What makes atoms different is the number of protons in the nucleus
• Each atom has its own number of protons in its nucleus
• The number of protons in its nucleus is called its atomic number
• Atoms are neutral because they have the same number of elecrons and protons
• For example if carbon is atom number 6 it has 6 protons and 6 electrons
• The electron configuration tells us how the electrons are arranged in an atom
• The first energy level holds 2 electrons, the second can hold up to 8 electrons, the third can hold up to 8 electrons and so can every other energy level after that
• For example an atom of Nitrogen has 7 electrons, 2 go in the first energy level and there are 5 left over so these go in the next energy level, its electron
configuration is 2, 5
• For example an atom of Potassium has 19 electrons, 2 go in the first energy level and there are 17 left over so 8 of these go in the next energy level, This leaves
9 electrons, 8 of these go in the next energy level which leaves 1 electron this go in the last energy level, its electron configuration is 2,8,8,1
• Some atoms have different numbers of neutrons in their nucleus even though they are the same atom, these are called isotopes
• Isotopes are atoms of the same element with different numbers of neutrons